Antimicrobial Treatment Options In the Management Of Odontogenic Infections
G.K.B. Sandor, DDS, MD, FRCD(C), FRCS(C), FACS
D.E. Low, MD, FRCP(C)
P.L. Judd, B.Sc., DDS, M.Sc., Dip. Pedo.
R.J. Davidson, PhD
Most acute orofacial infections are of odontogenic origin. In normal hosts, however, they usually do not occur without some type of predisposing condition. Early recognition and management of acute orofacial infections is critical, because rapid systemic involvement can occur, especially in children. Antimicrobial therapy has an essential role in the management of these infections. If it is initiated before surgery, it can shorten the period of infection and minimize associated risks. The etiology of odontogenic infections is usually attributed to the endogenous flora of the mouth, and not to the introduction of non-resident bacteria. Odontogenic infections are typically polymicrobial; however, anaerobes generally outnumber aerobes by at least four fold. The penicillins have historically been used as the first-line therapy in these cases, but increasing rates of resistance have lowered their usefulness. Bacterial resistance to this class of agents is predominately achieved through the production of ß-lactamases. Clindamycin, because of its broad spectrum of activity and resistance to ß-lactamase degradation, is an attractive first-line therapy in the treatment of odontogenic infections.
Introduction | Methods | Odontogenic Infections | Microbiology | Antimicrobial Therapy | Summary | Acknowledgments | References ]
Most acute orofacial infections are of odontogenic origin.1 The majority of odontogenic infections are self limiting, and may drain spontaneously. However, these infections may drain into the anatomical spaces adjacent to the oral cavity and spread along the contiguous fascial planes, leading to more severe infection.1 Due to the proximity of the central nervous system and critical respiratory passages, timely efforts are required to establish a patent airway, mechanical debridement and drainage, and appropriate antimicrobial therapy.2 This article will focus on the etiology of odontogenic infections, and the antimicrobial therapy used to treat them.
The content of this review was discussed during a meeting of specialists in pediatric dentistry, oral and maxillofacial surgery, infectious diseases, and microbiology (Toronto, 1997). Current information concerning the etiology, antimicrobial treatment, and antimicrobial resistance of odontogenic infections from 1990 to the present was obtained using a MEDLINE search. Key search phrases included odontogenic and infection, etiology, treatment, antibiotics, and resistance. Expert opinion and observations concerning the changing behavior of odontogenic infections were included where published data was lacking.
Odontogenic infections are among the most common infections of the oral cavity. They can be caused by dental caries, deep restorations that approximate the pulp chamber, pulpitis, periapical abscess, periodontitis, periodontal abscess, and pericoronitis. Odontogenic infections may develop into osteoperiostitis of the jaw, osteomyelitis, and deep fascial space infections. In normal hosts, acute odontogenic infections usually do not occur without some type of predisposing condition, such as periodontal accumulations, necrotic pulp tissue, or tissue damage associated with trauma or surgery.
The increasing rates of antimicrobial resistance are well recognized and described in the literature. However, there is some evidence to suggest that virulence has also changed in some bacteria.3 The epidemic of Brazilian purpuric fever in 1990, which was associated with a new clonal variation of Haemophilus influenzae, and the more recent devastating invasive infections caused by group A Streptococcus provide convincing evidence of this.3
There has also been a notable change in the behavior of odontogenic infections at our institutions. The inappropriate and increased use of resin restorations in the pediatric population has caused severe odontogenic infections to become an apparent and growing problem in this group.4 Furthermore, the severity of these infections in the adult population is far greater than in the past, with a more rapid and dramatic spread through the fascial planes surrounding the airways. This change has necessitated the use of CT scans to diagnose and determine the extent of severe odontogenic infections, as well as the more frequent use of a lifesaving surgical airway such as a tracheotomy, and broader incision and drainage techniques. The length of hospitalization for patients with these infections has also increased. Late referral makes the treatment of such patients more precarious. Dentists must therefore be prepared to refer their patients in a timely and appropriate manner.
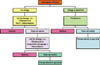
It is important for the dentist to develop an algorithm, or organized approach, when dealing with odontogenic infections (Fig. 1).
Fig 1: Algorithm for selecting antimicrobial therapy and referal to specialist. This approach should include the use of appropriate first-line antimicrobial therapy, and timely referral to a specialist if the results of such therapy are less than ideal. This will minimize potentially catastrophic and lethal complications such as airway obstruction and/or mediastinitis.
Although severe odontogenic infections are surprisingly common in the young child, there is a paucity of papers on the management of this patient group.4 Early management of these infections is essential because of the rapid systemic involvement that can occur, especially in children. Consequently, all infections should be treated as emergent or at least urgent.
The microbial flora of the oral cavity contains a complex population of microorganisms. In fact, the oral cavity is unique in that it cannot be regarded as a single uni-form environment, and resident microorganisms can be cultured from most areas of the mouth. However, certain sites such as the tongue, tooth surfaces, and gingival crevice tend to favor colonization by specific microorganisms.
Odontogenic infections are usually attributed to the endogenous flora of the mouth, and not to the introduction of non-resident bacteria. An important feature of suppurative odontogenic infections is that they are typically polymicrobial in nature, with mixed aerobic and anaerobic bacteria present.7-9 However, the anaerobes generally outnumber the aerobic bacteria by a factor of three to four fold.2,5,6
The mixed aerobic-anaerobic composition of the bacteria involved in suppurative odontogenic infections is thought to be important in the pathogenesis of infection. Barclay reported that if bacteria involved in mixed odontogenic infections are isolated in pure culture and transferred to healthy animals, they are often incapable of producing disease.10 Thus, a synergistic interdependence be-tween aerobic and anaerobic bacteria is thought to be necessary for the development of infection. Essentially, the respiration of aerobic bacteria depletes the local environment of oxygen, creating an oxygen-poor, nutrient-rich habitat suitable for anaerobic growth. Once anaerobiosis is achieved, anaerobes proliferate, secreting toxins and enzymes that result in tissue destruction and abscess formation.
The predominant bacterial species isolated from odontogenic infections are listed in Table I. Streptococcus (anaerobic), Peptostreptococcus, Veillonella, Lactobacillus, and Actinomyces account for approximately 80 per cent of the total cultivable microflora in the healthy oral cavity.11,12 Streptococcus spp account for approximately 95 per cent of the aerobic and facultative organisms colonizing the oral cavity. Aerobic gram negative bacilli are uncommon in healthy adults, but occur more frequently in hospitalized patients and the elderly.12 The microflora's bacterial composition is slightly different in patients with healthy tissue and patients with dental caries, gingivitis, and periodontitis. Streptococcus mutans' role in dental caries has been well established.13,14 With gingivitis and periodontitis, the predominant subgingival flora shifts to a greater proportion of anaerobic gram negative bacilli.15 In patients with pericoronitis, the predominant species recovered are Peptostreptococci, Fusobacterium, and Bacteroides, while Prevotella intermedia, Fusobacterium, and Capnocytophaga are frequently recovered from periodontal abscesses.9,16,17
Heimdahl et al examined orofacial infections and correlated their clinical appearance with the observed microbial etiology.17 Anaerobic gram negative rods (Bacteroides, Prevotella, Fusobacterium) were more frequently isolated from patients with severe infections than from those deemed to have mild infections (p <0.05). Fusobacterium nucleatum was predominately associated with severe infections (p < 0.05).
Although bacteria play a major role in odontogenic infections, antimicrobials are not always warranted. For example, a draining abscess or a fistula containing a chronic infection usually only requires the extraction of the offending tooth. However, other disease processes, including acute periapical abscesses, periodontal abscesses, pericoronitis, and deep fascial space infections may require antimicrobial therapy. Antimicrobials must never be used as a replacement for appropriate surgical drainage and/or debridement, and should only be used as adjunctive therapy. The maintenance of an airway and abscess drainage is a condition sine qua non.12 However, antimicrobial therapy initiated soon after diagnosis and before surgery can shorten the period of infection and minimize associated risks such as bacteremia.12
Penicillins and Cephalosporins
Historically, the penicillins have been used as first-line agents in the treatment of odontogenic infections. The penicillins are bactericidal. Although their spectrum of activity is relatively narrow, it is appropriate for the treatment of odontogenic infections. The penicillins exert their antibacterial effect by inhibiting a number of bacterial enzymes (the penicillin binding proteins) that are essential for peptidoglycan synthesis.18 Peak serum levels are achieved one to two hours after ingestion. With all penicillins other than amoxicillin, food can delay absorption and result in lower peak serum levels.11,19 The major adverse effects are hypersensitivity reactions, which can range in severity from a rash to immediate anaphylaxis. Anaphylactic reactions are extremely uncommon, however, the mortality rate is 0.001 per cent .11
Oral penicillin G is not recommended as empiric therapy for the treatment of odontogenic infections. Gastric acid inactivates the drug, with the result that only 30 per cent of the oral dose is absorbed.11
Phenoxymethylpenicillin (penicillin V) is the penicillin of choice for odontogenic infections. It has higher acid stability than penicillin G, and produces plasma levels two to five times higher than an equivalent dose of penicillin G.
Pivampicillin, the pivaloyloxymethyl ester of ampicillin and amoxicillin, and a broad-spectrum semisynthetic penicillin, offers no advantage over penicillin V in treating odontogenic infections. Pivampicillin and amoxicillin are, however, better absorbed orally than penicillin V.
Clavulin, a combination of amoxicillin and the ß-lactamase inhibitor clavulanic acid, retains activity against the ß-lactamase producing organisms commonly associated with odontogenic infections. Diarrhea is the major side effect of amoxicillin-clavulanic acid.
The first and second generation oral cephalosporins, while having a significantly broader spectrum of activity than the penicillins, do not offer any advantage over penicillin V in treating odontogenic infections. This is due to the principle odontogenic pathogens.
For prophylaxis against the endocarditis associated with dental procedures, the American Heart Association considers amoxicillin to be the first choice for non-allergic patients.19
Increasing rates of penicillin resistance and treatment failures have been reported.21-26 The highest rates of penicillin resistance have been observed with the members of the genus Bacteroides and Prevotella.21-23 Penicillin resistance in these pathogens has been correlated with ß-lactamase production. Heimdahl et al reported on a series of patients with orofacial infections who failed to respond to penicillin therapy due to ß-lactamase producing Bacteroides.24 Witcher et al described a series of patients who, after treatment with penicillin, developed mandibular osteomyelitis caused by ß-lactamase producing P. melaninogenicus.26 Using an animal model, ß-lactamase production by strains of P. melaninogenicus in a mixed infection has been shown to protect both P. melaninogenicus and other bacteria from penicillin.27 Due to the increasing prevalence of penicillin resistance, the Sanford Guide to Antimicrobial Therapy recently replaced penicillin V with clindamycin as the drug of choice in treating odontogenic infections.28
Macrolides
In Canada, the currently available macrolides include erythromycin, clarithromycin, and azithromycin. Macrolides are bacteriostatic agents that inhibit bacterial RNA-dependent protein synthesis.18 Erythromycin has adequate activity against the majority of odontogenic pathogens, but up to 50 per cent of Fusobacterium are resistant to erythromycin.19,29 The major side effect of erythromycin is gastric upset, but various esters (estolate, ethylsuccinate) and enteric-coated preparations are available that reduce gastric upset. Erythromycin crosses the placenta, reaching fetal serum concentrations of five to 20 per cent of maternal serum levels. The use of erythromycin in pregnancy is considered relatively safe, although erythromycin estolate should be avoided in pregnant women.30
In the United States, the Food and Drug Administration (FDA) classifies the use of macrolides during pregnancy as risk category B. No human trials have been performed, but the results of animal studies using drugs in this category have been inconsistent. Some have demonstrated no fetal risk, while others have demonstrated a risk. The newer macrolides, clarithromycin and azithromycin, offer improved pharmacokinetics compared to erythromycin. However, the macrolides should not be considered as first-line therapy in treating odontogenic infections, and should be reserved for patients with penicillin allergy.
Tetracyclines
The tetracyclines are of limited use in the practise of general dentistry. Although they are active against a range of both gram positive and gram negative aerobic and anaerobic bacteria, their usefulness has been greatly diminished by widespread resistance.
Doxycycline and minocycline possess better anaerobic activity than tetracycline, but neither of these agents should be considered first-line therapy for odontogenic infections.31 Gastrointestinal disturbances are common with the tetracyclines, and hypersensitivity reactions such as skin rash and drug fever can occur. The use of tetracyclines in children under 13 years of age and pregnant women is contraindicated due to the risk of tooth discoloration and interference with bone development.11
Metronidazole
Metronidazole is a bactericidal agent that is highly active against most anaerobes, but it lacks activity against aerobic bacteria. Similarly, although it retains activity against penicillin-resistant anaerobic gram negative bacilli, it only has moderate activity against microaerophilic gram positive cocci. In serious infections, metronidazole is best used in conjunction with penicillin to ensure coverage against aerobic gram positive bacteria. However, a combination of two drugs with different dosing schedules may lower patient compliance. The development of resistance to this agent by common odontogenic pathogens is rare.
Rapid absorption occurs after oral administration. Absorption is not affected by food, although peak serum levels may be delayed. Metronidazole is well tolerated. Mild side effects include gastrointestinal disturbances, a metallic taste, and an innocuous darkening of the urine.32 Patients should be warned not to consume alcohol while on this medication, as an "Antabuse" reaction (nausea, vomiting, abdominal cramps) can occur. Metronidazole crosses the placental barrier and should be withheld during the first trimester. The use of metronidazole during the second and third trimesters is not recommended, but can be considered if the potential benefits are weighed against possible risks.30 Clinical experience with metronidazole in the pediatric population is still limited. Metronidazole's excellent anaerobic gram negative activity, coupled with its low degree of toxicity, make it a good candidate in the treatment of some odontogenic infections.
Clindamycin
Clindamycin has excellent activity against gram positive organisms, including anaerobes and ß-lactamase producing strains. Low concentrations of the drug are bacteriostatic, but bactericidal activity is achieved clinically with the usual recommended doses. Clindamycin binds to the 50S ribosomal subunit of suscep-tible bacteria and interferes with protein synthesis.18 Over
90 per cent of clindamycin is absorbed following oral administration. Absorption is delayed but not decreased with the ingestion of food. The principle side effect associated with clindamycin is diarrhea, with a reported incidence ranging from 0.1 per cent to 17 per cent.6 Clindamycin has also been associated with pseudomembranous colitis. This concern is exaggerated, however, and the risk is probably no greater than with other broad spectrum antimicrobials.33 The broad spectrum penicillins and cepha-losporins are also frequently associated with this syndrome.34,35 However, until further clinical experience is obtained, clindamycin is not indicated in the newborn (infants below 30 days of age). The use of clindamycin in pregnant women is considered risk factor B.30
Clindamycin has excellent activity against aerobic gram positive cocci, such as Staphylococcus aureus, Streptococcus spp, and most anaerobes, including penicillin resistant strains of Bacteroides, Prevotella, and Porphyromonas spp.23
A number of clinical trials have demonstrated clindamycin's efficacy in treating odontogenic infections.25,36-39 Gilmore et al demonstrated comparable acti-vity between clindamycin and penicillin V in the treatment of moderate to severe odontogenic infections.36 Von Konow reported similar findings, but the clindamycin group had a shorter duration of fever, pain, and swelling.37
Clindamycin has been used successfully to treat patients when therapy with other agents has failed.25,39 Thirty patients with a history of unsuccessful treatment involving scaling, periodontal surgery, and tetracycline therapy were treated successfully with scaling and clindamycin.39 Heimdahl et al and Witcher et al reported on a series of patients failing penicillin therapy due to ß-lactamase producing P. melaninogenicus (Bacteroides).24,26 All were successfully treated with clindamycin.
Clindamycin's broad spectrum of coverage and excellent clinical efficacy, coupled with the increase in both penicillin resistance and the reports of treatment failures with penicillin, has prompted the Sanford Guide to Antimicrobial Therapy to replace penicillin V with clindamycin as the drug of choice in treating odontogenic infections.28
Several studies have demonstrated clindamycin's efficacy in preventing dry socket.40,41 Kupfer reported that clindamycin significantly reduced the incidence of dry socket following the removal of impacted mandibular third molars compared to three controls, lincomycin, penicillin V, and furacin powder.40 Trieger and Schlage also demonstrated that clindamycin, embedded in gelfoam, was significantly more effective in preventing dry socket than placebo-treated sockets.41
Recently, the American Heart Association recommended giving clindamycin, rather than erythromycin, to penicillin-allergic patients requiring endocarditis prophylaxis.20
Fluoroquinolones
The fluoroquinolones currently available in Canada are ciprofloxacin, norfloxacin, oflo-xacin, and levofloxacin. These agents are bactericidal, and exert their antibacterial effect by inhi- biting DNA gyrase and topoisomerase IV.42 The fluoroquinolones have potent gram negative acti-vity, including activity against Pseudomonas spp. Their activity against gram positive bacteria is marginal, however.42 Due to the spectrum of organisms associated with odontogenic infections, the use of fluoroquinolones in the treatment of acute odontogenic infections should not be considered.
Oral Versus Intravenous Antibiotics
Most infections, when treated in a timely matter, can be adequately managed using oral therapy. Patients with no airway swelling, eyelid swelling or neck involvement, who have normal oral intake and systemically feel well, are good candidates for oral therapy. However, intravenous antibiotics and hospital admission should be strongly considered when swelling of the airway, swelling of the eyelid, or neck involvement is present, or the patient's level of activity and oral intake is decreased. The presence of airway or eyelid swelling and neck involvement also warrants a diagnostic CT scan to avoid potentially disastrous complications.
Odontogenic infections are typically polymicrobial. The pathogenesis of odontogenic infections is dependent on a synergistic relationship between aerobic and anaerobic bacteria.
The last decade has seen a notable change in the behavior of odontogenic infections. The severity of these infections is far greater than in the past, demanding swift recognition of the disease followed by prompt, more aggressive treatment. Failing to identify and treat these infections promptly may result in disastrous outcomes, as occurred in a recently sensationalized case entitled "Death of a Toothache Victim," which was reported in March 1998 in the Toronto Star.43
Historically, the penicillins have been used as first-line agents in the treatment of odontogenic infections. The increasing prevalence of ß-lactamase production in dental pathogens and the coincident reports of clinical failures have decreased the usefulness of these agents, making clindamycin more attractive. Amoxicillin, however, is still the drug of choice for prophylaxis against endocarditis.
Clindamycin has been increasingly tested for use in a variety of dental infections. Even in situations where penicillin has failed, clindamycin's excellent activity against both aerobic gram positive cocci and anaerobic pathogens make this agent well suited in the treatment of odontogenic infections.
Dr. Sandor is coordinator of oral and maxillofacial surgical services at The Hospital for Sick Children and the Bloorview-MacMillan Centre; acting head of oral and maxillofacial surgery at the Toronto Hospital, and assistant professor, faculty of dentistry, University of Toronto.
Dr. Low is microbiologist-in-chief at the Mount Sinai and Princess Margaret Hospitals, and professor, faculty of medicine and department of microbiology, University of Toronto.
Dr. Judd is the director, division of pediatric dentistry, The Hospital for Sick Children and assistant professor, faculty of dentistry, University of Toronto.
Dr. Davidson is chief microbiology resident, departments of microbiology, University of Toronto and Mount Sinai Hospital.
Reprint requests to: Dr. George K.B. Sandor, Department of Dentistry, Hospital for Sick Children, 555 University Ave., Toronto ON M5G 1X8
- Heimdahl, A. and Nord, C.E. Treatment of orofacial infections of odontogenic origin. Scand J Infect Dis 46(Suppl):101-105, 1985.
- Baker, K.A. and Fotos, P.G. The management of odontogenic infections. A rationale for appropriate chemotherapy. Dent Clin North Am 38:689-706, 1994.
- Morse, S.S. Factors in the emergence of infectious diseases. Emerg Infect Dis 1:7-15, 1995.
- Judd, P.L. and Sandor, G.K.B. Management of odontogenic orofacial infection in the young child. Ont Dent 74:39-45, 1997.
- Sands, T., Pynn, B.R. and Katsikeris, N. Odontogenic infections: Microbiology, antibiotics, and management. Oral Health 85:11-28, 1995.
- Sands T. and Pynn, B.R. Odontogenic infections and clindamycin. UTDJ 32-33, 1995.
- Greenberg, R.N., James, R.B., Marier, R.L. et al. Microbiologic and antibiotic aspects of infections in the oral and maxillofacial region. J Oral Surg 37:873-884, 1979.
- Kannagara, D.W., Thadepalli, H. and McQuirter, J.L. Bacteriology and treatment of dental infections. Oral Surg 50:103-109, 1980.
- Von Konow, L., Nord, C.E. and Nordenram, A. Anaerobic bacteria in dentoalveolar infections. Int J Oral Surg 10:313-322, 1981.
- Barclay, J.K. Antibiotics revisited. N Z Dent J 86:44-47, 1990.
- Chow, A.W. Infections of the oral cavity, neck, and head. In: Mandell, G.L., Douglas, R.G., Bennett, J.E., eds. Principles and Practice of Infectious Diseases. 3rd ed. New York, NY. Churchill Livingstone Inc. 1990.
- Ehrenfeld, M. Clindamycin in the treatment of dental infections. In: Zambrano, D., ed. Clindamycin in the Treatment of Human Infections. Kalamazoo, Michigan. Upjohn Company, 1992.
- Hamada, S. and Slade, H.D. Biology, immunology, and carcinogenicity of Streptococcus mutans. Microbiol Rev 44:331-384, 1980.
- Loesche, W.J. Role of Streptococcus mutans in human dental decay. Microbiol Rev 50:353-380, 1986.
- Lo Bue, A.M., Sammartino, R., Chisari, G. et al. Efficacy of azithromycin compared with spiramycin in the treatment of odontogenic infections. J Antimicrob Chemother 31(Suppl E):119-127, 1993.
- Newman, M.G. Anaerobic oral and dental infection. Rev Infect Dis 6:S107-S113, 1984.
- Heimdahl, A., Von Konow, L., Satoh, T. et al. Clinical appearance of orofacial infections of odontogenic origin in relation to microbiological findings. J Clin Microbiol 22:299-302, 1985.
- Yao, J.D.C. and Moellering, R.C. Antibacterial agents. In: Murry, P.R., Baron, E.J., Pfaller, M.A. et al, eds. Manual of Clinical Microbiology. 6th ed.; ASM Press, Washigton D.C. 1995.
- Karlowsky, J., Ferguson, J. and Zhanel, G. A review of commonly prescribed oral antibiotics in general dentistry. J Can Dent Assoc 59:292-294,297-300, 1993
- Dajani, A.S., Taubert, K.A., Wilson, W. et al. Prevention of bacterial endocarditis: Recommendations by the American Heart Association. JAMA 277:1794-1801, 1997.
- Murray, P.R. and Rosenblatt, J.E. Penicillin resistance and penicillinase production in clinical isolates of Bacteroides melaninogenicus. Antimicrob Agents Chemother 11:605-608, 1977.
- Edson, R.S., Rosenblatt, J.E. and Lee, D.T. Recent experience with antimicrobial susceptibility of anaerobic bacteria: increasing resistance to penicillin. Mayo Clin Proc 57:737-741, 1982.
- Kinder, S.A., Holt, S.C. and Korman, K.S. Penicillin resistance in subgingival microbiota associated with adult periodontitis. J Clin Microbiol 23:1127-1133, 1986.
- Heimdahl, A., Von Konow, L. and Nord, C.E. Isolation of ß-lactamase producing Bacteroides strains associated with clinical failures with penicillin treatment of human orofacial infections. Arch Oral Biol 25:689-692, 1980.
- Walker, C. and Gorden, J. The effect of clindamycin on the microbiota associated with refractory periodontitis. J Periodontol 61:692-698, 1990.
- Witcher, B.L., Beirne, O.R. and Smith, R.A. ß-lactamase producing Bacteroides melaninogenicus and osteomyelitis of the mandible. J Oral Med 38:17-20, 1983.
- Hackman, A.S. and Wilkins, T.D. Influence of penicillinase production by strains of Bacteroides melaninogenicus and Bacteroides oralis on penicillin therapy of experimental mixed anaerobic infection in mice. Arch Oral Biol 21:385-389, 1976.
- Sanford, J.P., Gilbert, D.N. and Moellering, R.C. Selection of initial empirical antimicrobial therapy on clinical grounds. In: Sanford, J.P., Gilbert, D.N., Moellering, R.C., eds. The Sanford Guide to Antimicrobial Therapy. 1996.
- Peterson, L.J. Microbiology of head and neck infections. Oral Maxillofac Surg Clin North Am 3:247, 1991.
- Dashe, J.S. and Gilstrap, L.C 3rd. Antibiotic use in pregnancy. Obstet Gynecol Clin North Am 3:617-629, 1997.
- Moellering, J.E., Nelson, C.L. and Kohler, R.B. The microbiology and chemotherapy of odontogenic infections. J Oral Maxillofac Surg 47:976, 1989.
- Slots, J. and Rams, T.E. Antibiotics in periodontal therapy: Advantages and disadvantages. J Clin Periodontol 17:479-493, 1990.
- Devenyi, A.G. Antibiotic induced colitis. Sem Ped Surg 4:215-220, 1995.
- McFarland, L.V., Surawicz, C.M. and Stamm, W.E. Risk factors for Clostridium difficile carriage and C.difficile associated diarrhea in a cohort of hospitalized patients. J Infect Dis 162:678-684, 1990.
- Devenyi, A.G. Antibiotic induced colitis. Sem Ped Surg 4:215-220, 1995.
- Gilmore, W.C., Jacobus, N.V., Gorbach, S.L. et al. A prospective double blind evaluation of penicillin versus clindamycin in the treatment of odontogenic infections. J Oral Maxillofac Surg 46:1065-1070, 1988.
- Von Konow, L., Kondell, P.A., Nord, C.E. et al. Clindamycin versus phenoxymethylpenicillin in the treatment of acute orofacial infections. Eur J Clin Microbiol Infect Dis 11:1129-1135, 1992.
- Mangundjaja, S. and Hardjewinata, K. Clindamycin versus ampicillin in the treatment of odontogenic infections. Clin Ther 12:242-249, 1990.
- Gordon, J., Walker, C., Hovliaras, C. Efficacy of clindamycin hydrochloride in refractory periodontitis: 24-moth results. J Periodontol 61:686-691, 1990.
- Kupfer, S.K. Prevention of dry socket with clindamycin. NYSDJ. June/July:30-36, 1995.
- Trieger, N. and Schlagel, G.D. Preventing dry socket. A simple procedure that works. JADA 122:67-68, 1991.
- Drlica, K. and Zhao, X. DNA gyrase, topoisomerase IV and the 4-quinolones. Microbiol Mol Biol Rev 61:377-392, 1997.
- Daly, R. Death of a toothache victim. The Toronto Star. March 1st, A1,A14, 1998.